- Product Listings
- Compare Products
- Calculators
- Product Tours
- Expert Reviews
- Our Experts
- Company Directory
- About Beye
- Contact Us
Ⓒ 2025 Beye.com. All rights reserved.
This content is intended for health care professionals and providers only. The information contained on Beye.com, including text, graphics, images, and interactive activities, is for informational purposes only, and is not intended to be a substitute for professional medical advice. Beye LLC, via its Editors and Publisher, accepts no responsibility for any injury or damage to persons or property occasioned through the implementation of any ideas or use of any product described herein. Although great care is taken to ensure that all information is accurate, it is recommended that readers seek independent verification of advice on drugs and other product usage, surgical techniques and clinical processes prior to their use. References made in article may indicate usage of medical equipment or drugs at dosages, for periods of time, and in combination not included in the current prescribing information. Inclusion of advertising materials on the website thereof, does not constitute and representation or guarantee by Beye LLC of the quality of such products, or of the claims made.
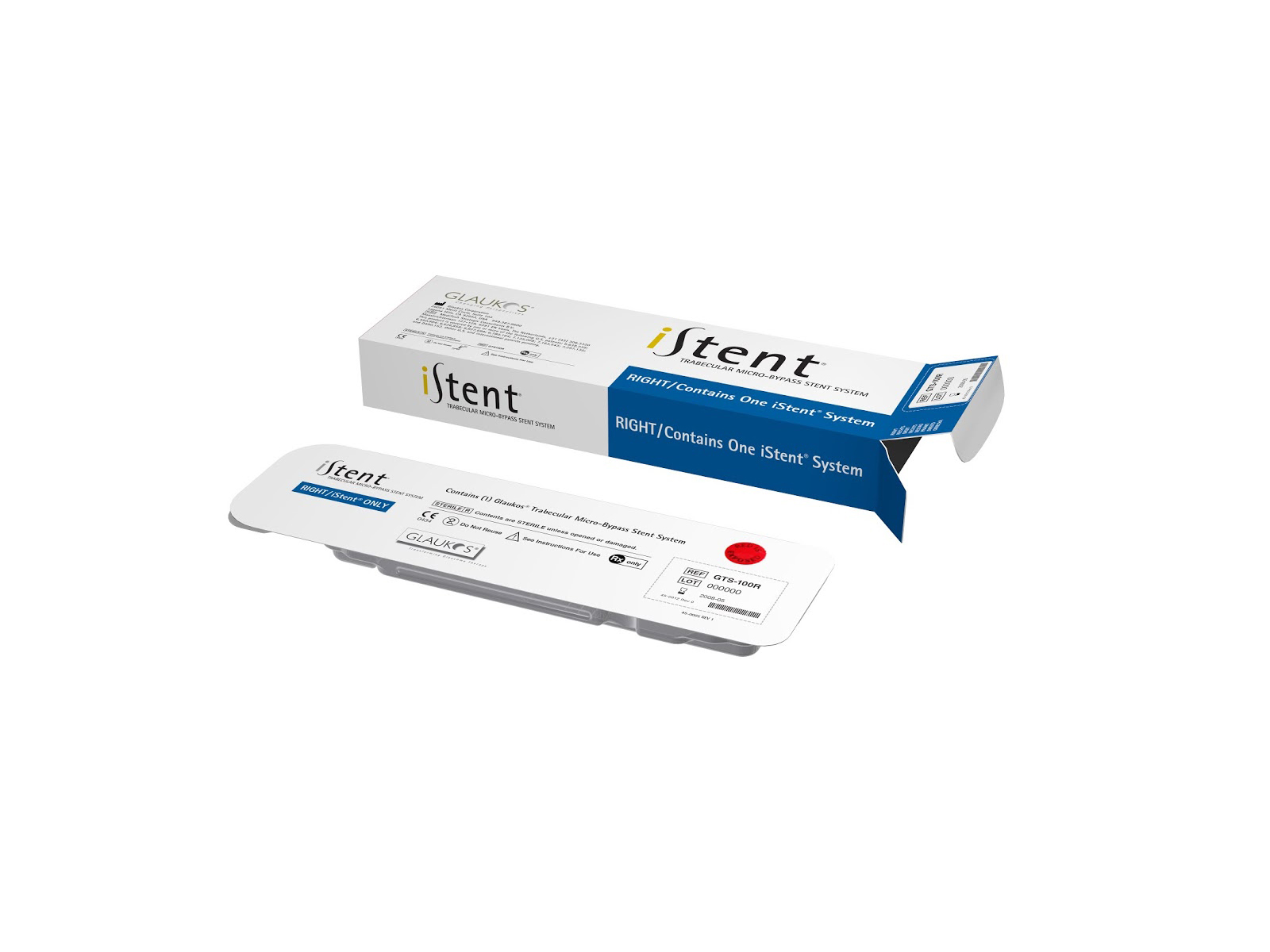
iStent Supra Micro-Bypass Stent
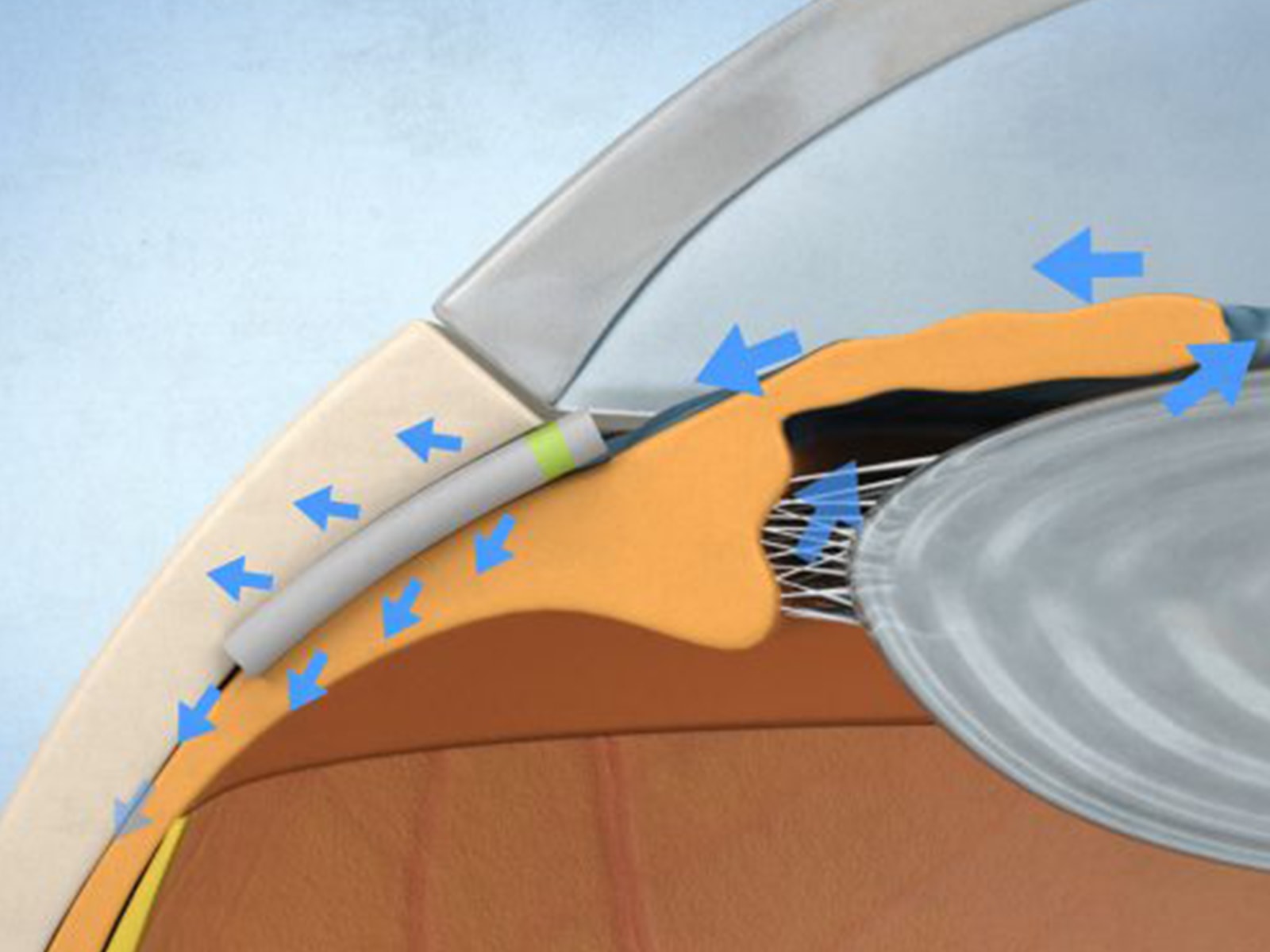
GlaukosThe iStent Supra is designed to reduce IOP by accessing the suprachoroidal space in the eye. Approximately 4.0 mm in length and curved to follow the eye?s anatomy, the iStent Supra is already approved for marketing in the European Union and certain other countries outside the United States.
At-a-Glance
- Material: Polymer
- Ridged tube designed to improve implant retention in its desired location
Details
FDA
Yes
CE Mark
Yes
Classification
Ab interno
Indication
Not specified
Mechanism of Action
Creates a channel to enable aqueous outflow to the suprachoroidal space
Usage
Treatment of mild to moderate glaucoma during cataract surgery or as a stand-alone procedure
Company Information
Contact the company for additional information, availability, or pricing:
Glaukos
glaukos.com229 Avenida Fabricante
San Clemente, CA 92672
Trending in Glaucoma
Powered by:
On the Front Lines of MIGS
Andrew A. Kao, MD
MillennialEye, May/June '18
Glaucoma Research Highlights
MillennialEye, May/June '18
Davinder S. Grover, MD, MPH
Davinder S. Grover, MD, MPH
CRSToday, April 2018
Pseudophakic Bullous Keratopathy
CRSToday, April 2018
MIGS: A Crash Course
Constance O. Okeke, MD, MSCE
CollaborativeEye, Mar/Apr '18
MIGS: The New Glaucoma Paradigm
Robert S. Stutman, OD, MBA, FAAO
CollaborativeEye, Mar/Apr '18
Persistent LASIK Flap Interface Fluid After DSAEK Procedure
CRSToday, February 2018
MIGS Update
CRSToday, Sep 2016
Show More